Theory
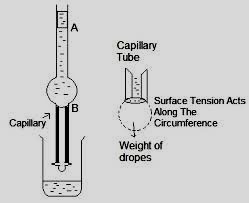
For a liquid, surface tension is defined as the force acting at right angles to the line of length, one centimeter, drawn on the surface of the liquid. It is usually represented by the Greek letter γ and its unit is dynes/cm. The surface tension may be measured relative to the pure liquid by using stalagmometer, which operates on ‘drop weight’ principle. Stalagmometer has a bulb with an attached capillary and has marks on both the sides of the capillary. Now suck the volume of liquid up to the upper mark, and allow it to release drop wise up to the lower mark, in this manner volume of liquids is fixed. The size of the drop of liquid forming on at the end of capillary depends upon the surface tension of the liquid. The drop falls when the total surface (i.e., 2πrγ) of the drop is equal to its weight (i.e. w = mg = 2πrγ).
For liquids
Since volume is fixed and n1 and n2 is the number of drops formed by volume v of the two liquids, then their surface tensions are related as



Applications:
1. Residential Water Softening
2. Laundry Facilities
3. Dishwashing
4. Boiler Feedwater Treatment
5. Cooling Tower Water Treatment
