Introduction
Adsorption is a surface phenomenon of solids, it occurs due to the presence of residual or unbalanced forces
exist with the surface molecules of the solids. These surface molecules attract the molecules/ions of other
species that come in their contact in either from i.e., in gas or in solution phase. This adhesion of
molecules/ ions to the surface is known as adsorption, it is of two types one is physical adsorption and the
other one is chemical adsorption based on the forces that bind these foreign species to the surfaces
molecules. It depends on following factor-
i) Specific area of the solid
ii) Solute concentration in solution at equilibrium
iii) Pressure in case of gaseous adsorption
iv) Nature of adsorbent
v) Temperature
There is a greater concentration of the adsorbed molecules at the surface of the solid as compared to the gas
phase or solution. The adsorption of a solute from a solution generally follows the Freundlich empirical
adsorption isotherm, given by the formula:
On taking log on both sides
Where,
x = weight of adsorbent
m = mass of the adsorbent
k = proportionality constant representing the capacity of the adsorbent
c = equilibrium concentration of the solution
If we draw the plot for Eq. 1 and 2 we get the following type of graphs:
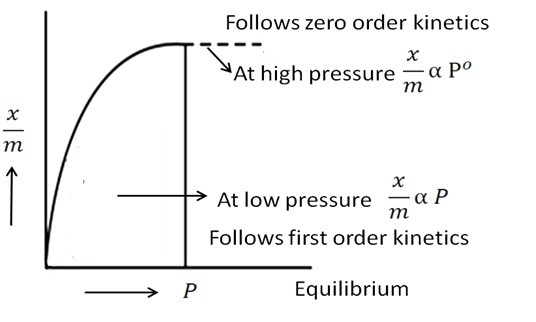
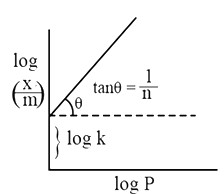
Applications:
1. Water Treatment Optimization
2. Environmental Monitoring
3. Industrial Processes
4. Drinking Water Quality
5.Research and Education
