Introduction
Pure water is neutral in nature with pH 7. Due to the presence of dissolved minerals in rain water, the pH increases and becomes alkaline. Alkaline nature of water is due to the presence of hydroxide (OH-), carbonate (CO32-) and bicarbonate (HCO3-) ions. The alkalinity contributed in water by these ions is estimated by titrating the water sample against a standard acid solution using phenolphthalein and methyl orange indicators successively. On reaction with acid hydroxide ions are completely neutralized to water and the end point for this reaction is determined by phenolphthalein indicator (one step neutralization)
Since neutralization of carbonate ions is a two steps reaction. In the first step, carbonate ions are neutralized to bicarbonate ions and completion of this step is determined by phenolphthalein indicator (pH 8.3-10). The bicarbonates thus formed further reacts with acid and neutralized completely to water and carbon dioxide in the second step and for this step end point is determined by methyl orange indicator (pH 3.1-4.4).
Further the alkalinity due to bicarbonate ions is neutralized completely in a single step and completion of this reaction is determined by methyl orange indicator.
These three reactions can be summarized as shown below:
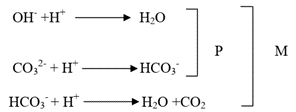
Here P and M represents alkalinity determined by phenolphthalein and methyl orange indicators, respectively. It is important to note that in water hydroxide and bicarbonate ions cannot exist together because both reacts with each other according to the following reaction:-
So there exists five different possibilities of alkalinity in water on the basis of concentration of various ions in water as tabulated in the table-
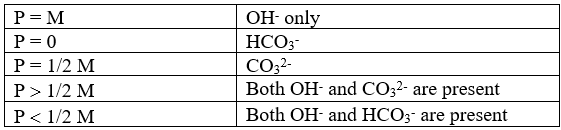
Here P = alkalinity measured by phenolphthalein end point
M = alkalinity measured by methyl orange end point
Applications:
1. Water Treatment Optimization
2. Environmental Monitoring
3. Industrial Processes
4. Drinking Water Quality
5.Research and Education
