Introduction
Hardness of water is a measure of amount of calcium (Ca2+) and magnesium (Mg2+) salts dissolved in water.
Hardness has also been discussed in terms of carbonate (temporary) and non-carbonate (permanent) hardness.
Carbonate hardness refers to the amount of carbonates and bicarbonates present in water that can be
precipitated or removed simply by boiling and therefore termed as temporary hardness. This type of hardness is
responsible for the deposition of scale in boilers and pipes for hot water. Non-carbonate hardness is caused
by salts of calcium and magnesium with sulphate, chloride or nitrate ions this type of hardness can’t be
removed by boiling therefore, referred as permanent hardness.
Though there is no health hazards associated with hardness of water but it causes scale and reduces the
lathering of soaps. Hardness of water is expressed in term of CaCO3 in ppm (i.e., mg/L). On the scale of CaCO3
equivalent concentration (in ppm), drinking water has been classified as soft water (0-60 ppm), medium 60-120
ppm, hard 120-180 ppm and very hard > 180 ppm.
Among various methods for determining the hardness of water complexometric titration method is easy to perform
in the laboratory. In this method disodium salt of EDTA (Ethylenediaminetetraaceticacid) forms colorless
stable complexes with Ca2+ and Mg2+ ions present in the water sample. Eriochrome Black-T (EBT), a dye is used
as an internal indicator, when 2-3 drops of EBT are added to the hard water it forms wine red colored unstable
complex with Ca+2 and Mg+2 ions around pH-10 (see reaction scheme). Basic buffer solution (NH4Cl + NH4OH) is
used to maintain the pH ~ 9-10 of the solution. This wine red-colored complex is titrated against standard
EDTA solution the color of the complex changes wine red to original blue color at the endpoint.
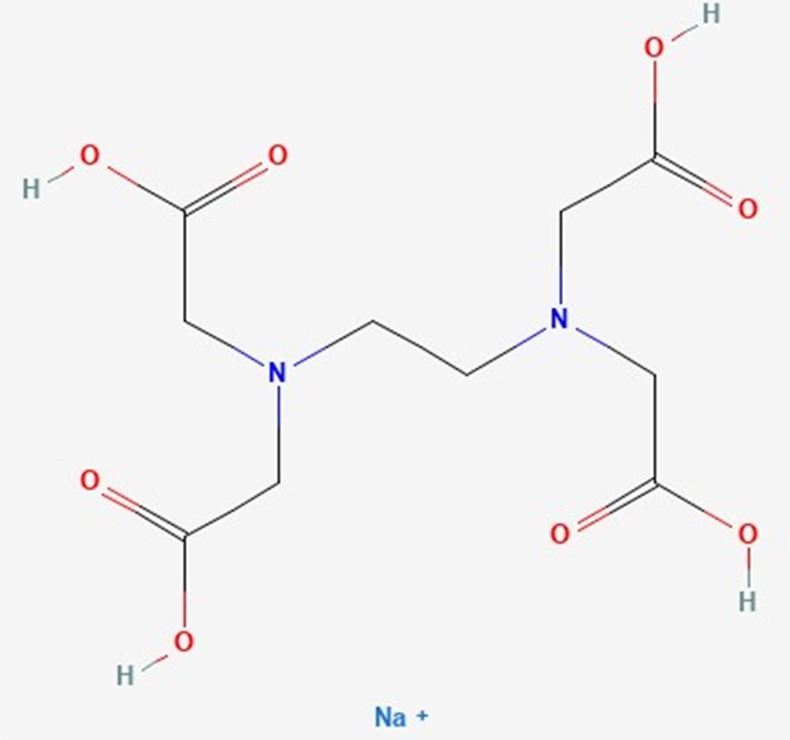
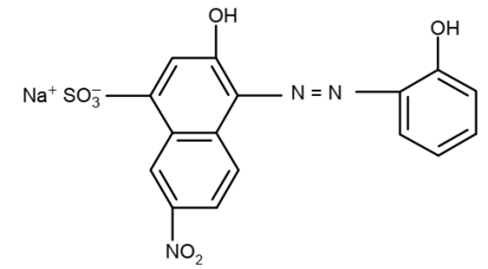
In aqueous solution EDTA ionizes to give 2Na+ ions and act as a strong chelating agent.



Applications:
1. Water Treatment Plants
2. Quality Control in Industries
3. Environmental Monitoring
4. Research and Education
5. Household Water Testing
