Introduction
Lead-acid and lithium-ion batteries are two of the most widely used battery technologies today. Each has its own strengths and weaknesses, making them suitable for different applications. This project aims to compare these batteries in terms of voltage, current, state of charge (SOC) using MATLAB's simulation and analysis capabilities. By exploring these key parameters, users can better understand how each battery type performs under various conditions.
1. Lead-Acid Battery
A lead-acid battery is a type of rechargeable battery that uses lead (Pb) and lead dioxide (PbO₂) electrodes submerged in sulfuric acid (H₂SO₄) electrolyte to store and release electrical energy. It is one of the oldest and most widely used battery technologies, known for its low cost, reliability, and ability to deliver high surge currents.
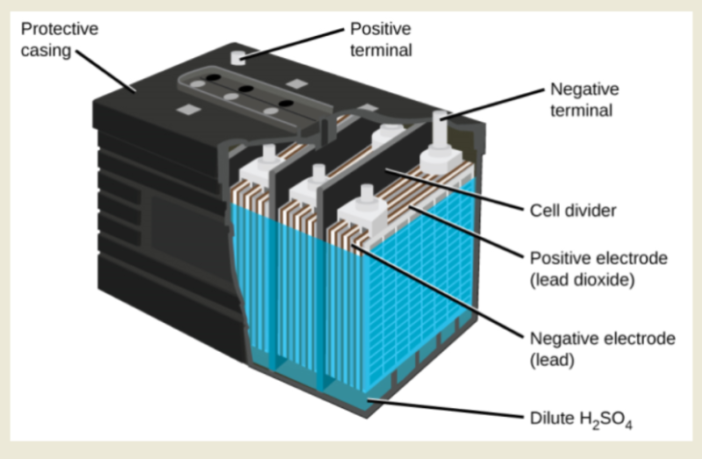
fig.1 : Lead-Acid Battery
Anode Reaction:
Pb + SO42- → PbSO4 + 2e-
Cathode Reaction:
PbO2 + 4H+ + SO42- + 2e - → PbSO4 + 2H2O
Overall Reaction:
Pb + PbO2 + 2H2SO4 → 2PbSO4 + 2H2O
Electrolyte
Sulfuric acid (H₂SO₄) which is diluted in water.
Working Principle:
During discharge, lead dioxide at the positive plate and lead at the negative plate react with sulfuric acid to form lead sulfate and water, releasing electrical energy. During charging, the process is reversed, converting lead sulfate back to lead, lead dioxide, and sulfuric acid.
Characteristics:
Heavier and bulkier than lithium-ion batteries. Lower energy efficiency and shorter lifespan. Shows a significant voltage drop when discharging.
2. Lithium-Ion Battery
A lithium-ion (Li-ion) battery is a rechargeable energy storage device that utilizes lithium ions to transfer charge between the positive (cathode) and negative (anode) electrodes. It is known for its high energy density, long lifespan, and fast charging capabilities, making it the preferred choice for modern electronic devices, electric vehicles (EVs), and renewable energy storage.
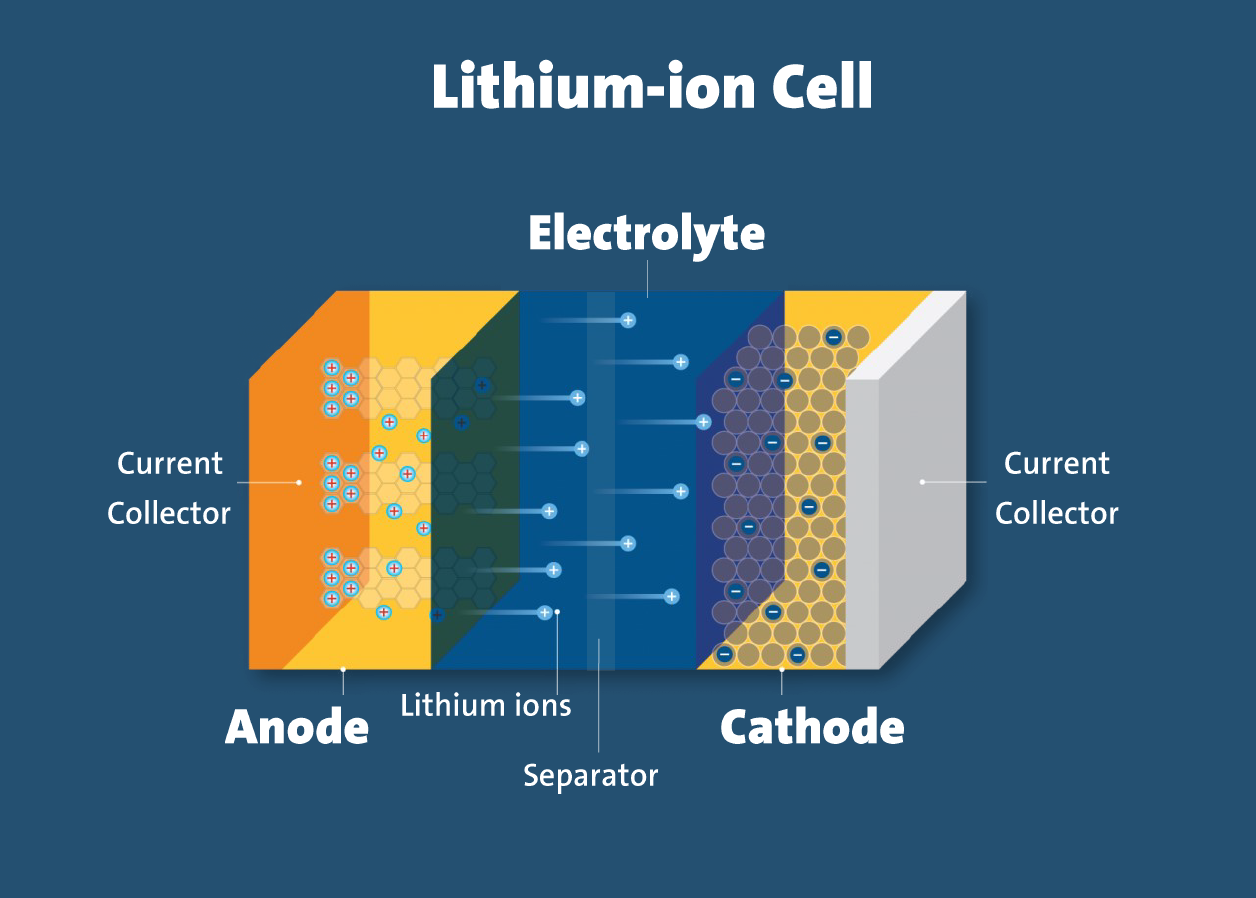
fig.2 : Lithium-Ion Battery
Anode Reaction:
LiC6 → Li + + e- + C6
Cathode Reaction:
LiCoO2 + Li+ + e- → Li2CoO2
Overall Reaction:
LiC6 + CoO2 → C6 + LiCoO2
Electrolyte:
Typically, a lithium salt (such as LiPF₆) dissolved in an organic solvent.
Working Principle:
During discharge, lithium ions move from the anode (typically graphite) to the cathode (typically a lithium metal oxide) through the electrolyte, generating electric current by releasing electrons. During charging, an external voltage source drives the lithium ions back to the anode.
Key Comparisons
| Parameter | Lead-Acid Battery | Lithium-Ion Battery |
|---|---|---|
| Voltage Stability | Drops gradually | Stays stable |
| Weight & Size | Heavy & bulky | Lightweight & compact |
| Energy Efficiency | Lower | Higher |
| Lifespan | Shorter | Longer |
| Charge/Discharge Rate | Slower | Faster |
Graph
1. Voltage Characteristics
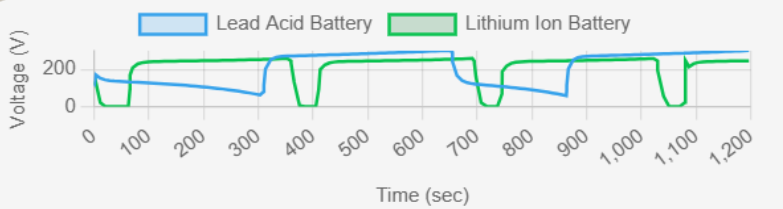
fig.3 : Voltage (V) vs Time (sec)
The black line likely represents one battery type (e.g., lead-acid), while the blue line represents the other (e.g., lithium-ion). Lithium-ion batteries maintain a stable voltage throughout the charge-discharge cycle, with a sharp cutoff at the end. Lead-acid batteries exhibit a more gradual voltage drop over time, which affects the efficiency of energy delivery. In the graph, you can see periodic fluctuations in voltage, suggesting charge-discharge cycles, with lithium-ion showing a more consistent voltage level compared to lead-acid. Key Observations from Graph
✔ Lithium-ion: Flat voltage curve, efficient energy utilization.
✔ Lead-acid: Gradual voltage drop, requiring frequent recharging.
2. State of Charge (SOC)
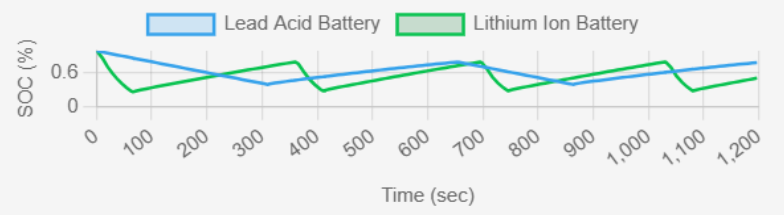
fig.4 : SOC (%) vs Time (sec)
The SOC graph shows a linear increase and decrease in both cases, but lithium-ion follows a more predictable pattern. Lead-acid batteries suffer from Peukert's effect, meaning their usable capacity decreases at higher discharge rates. Lithium-ion batteries provide a more accurate SOC estimate, while lead-acid SOC is influenced by discharge history. Key Observations from Graph
✔ Lithium-ion: More stable SOC fluctuations, supporting efficient charge-discharge cycles.
✔ Lead-acid: Steeper decline, meaning lower usable capacity over time.
3. Speed Characteristics
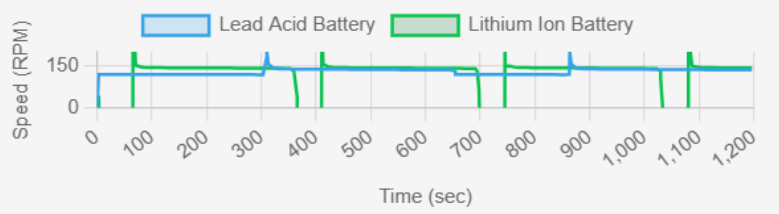
fig.5 : Speed (RPM) vs Time(sec)
Speed in battery performance refers to how quickly they respond to charge/discharge events. The lithium-ion battery shows quicker transitions in the charge/discharge cycle compared to lead-acid. Lead-acid batteries have a lag due to slower electrochemical reactions. Key Observations from Graph
✔ Lithium-ion: Faster response time, ideal for high-speed applications.
✔ Lead-acid: Slower reaction times, better suited for steady loads
4. Current Characteristics
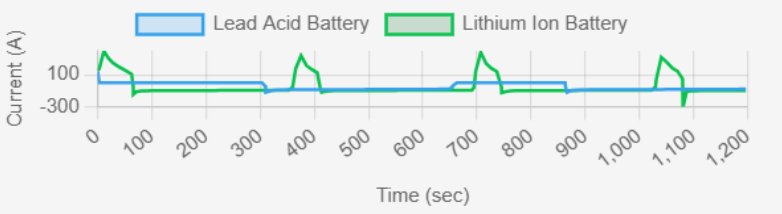
fig.6 : Current (A) vs Time (sec)
The current response of lithium-ion batteries is more stable and can handle rapid changes in demand. Lead-acid batteries show higher peaks and dips, indicating slower response to load variations. Lithium-ion batteries can handle higher discharge rates without major performance losses. Key Observations from Graph
✔ Lithium-ion: Fast response to load, minimal current fluctuations.
✔ Lead-acid: Slower response, higher variations.
Lithium-ion Battery (Advantages & Disadvantages)
Advantages:
• Higher energy density (more energy stored per unit weight)
• Lightweight and compact
• Longer lifespan (more charge-discharge cycles)
• Faster charging capability
• Low self-discharge rate
• Requires little to no maintenance
Disadvantages:
• Expensive compared to lead-acid batteries
• Sensitive to high temperatures and overcharging
• Requires protection circuits to ensure safe operation
• Environmental concerns in mining lithium and disposal
• Risk of fire or explosion if damaged
Lead-acid Battery (Advantages & Disadvantages)
Advantages:
• Lower cost and widely available
• Simple charging requirements
• Proven and mature technology
• Recyclable and environmentally manageable
• Tolerant to overcharging and mishandling
Disadvantages:
• Lower energy density (heavier for same power)
• Shorter lifespan and fewer charge-discharge cycles
• Requires regular maintenance (like checking electrolyte levels)
• Slower charging rate
• High self-discharge rate when idle
Applications
Lithium-ion Battery:
• Used in smartphones, laptops, and tablets
• Electric vehicles (EVs) and hybrid cars
• Power banks and portable electronic devices
• Solar energy storage systems
• Medical devices and aerospace equipment
Lead-acid Battery:
• Used in automobiles for engine starting
• Backup power for UPS systems
• Inverter and emergency lighting systems
• Industrial forklifts and wheelchairs
• Off-grid solar power systems
Conclusion
Lithium-ion batteries are more efficient, lighter, and have a longer lifespan compared to lead-acid batteries. However, lead-acid batteries are still used in some applications due to their lower cost and reliability.
