Theory
Hydrogen fuel cells produce electricity through a chemical reaction. Each cell consists of two electrodes: a negative anode and a positive cathode. The reaction that generates electricity occurs at these electrodes, with an electrolyte carrying charged particles between them and a catalyst accelerating the reactions.
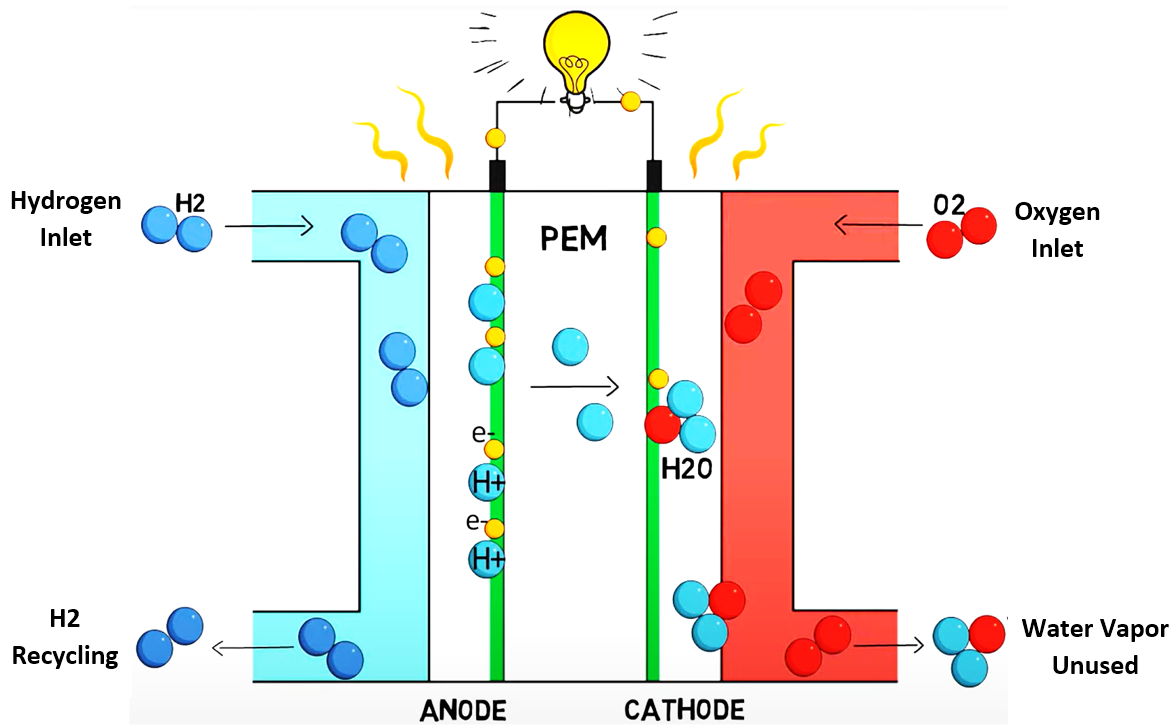
Fig. 1 Hydrogen Fuel Cell
Basic Process of a Fuel Cell
- Hydrogen enters at the anode, while oxygen enters at the cathode as shown in Fig.1.
- At the anode, hydrogen atoms split into protons and electrons.
- The positively charged protons pass through the electrolyte membrane to the cathode, while the electrons are diverted through a circuit, creating an electric current.
- At the cathode, protons and electrons combine with oxygen, forming heat and water as by-products.
In a fuel cell, the hydrogen combustion reaction is split into two electrochemical half reactions:
H2 → 2H+ + 2e −
½ O2 + 2H+ + 2e − → H2O
Since individual fuel cells generate only small amounts of electricity, they are combined in stacks to meet power requirements for various applications, from small devices to large-scale power plants.
Unlike batteries, fuel cells do not deplete or need recharging and can produce continuous electricity as long as they are supplied with fuel. With an anode, cathode, and electrolyte membrane and no moving parts, fuel cells are silent and highly reliable.
Fuel Cell Losses
Unfortunately, it is hard to maintain a high fuel cell voltage under current load. The voltage output of a real fuel cell is less than the thermodynamically predicted voltage output due to irreversible losses. The more current that is drawn from the cell, the greater these losses.
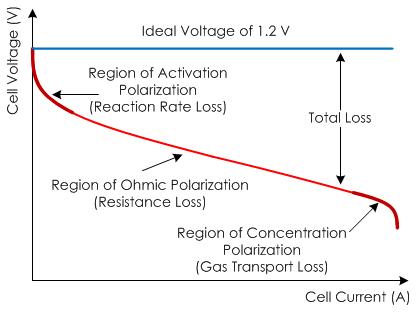
Fig. 2 Cell Voltage v/s Cell Current
There are three major types of fuel cell losses, which give a fuel cell V-I curve its characteristic shape:
- Activation losses (losses due to electrochemical reaction).
- Ohmic losses (losses due to ionic and electronic conduction).
- Concentration losses (losses due to mass transport).
The real voltage output for a fuel cell can be written as:
V = Ethermo − ηact − ηOhmic − ηconc
where:
- V = real output voltage of the fuel cell
- Ethermo = thermodynamically predicted fuel cell voltage output
- ηact = activation losses due to reaction kinetics
- ηOhmic = Ohmic losses from ionic and electronic conduction
- ηconc = concentration losses due to mass transport
The three major losses each contribute to the characteristic shape of the fuel cell V-I curve:
- Activation losses mostly affect the initial part of the curve.
- Ohmic losses are most apparent in the middle section of the curve.
- Concentration losses are most significant in the tail of the V-I curve.
Advantages of Hydrogen Fuel Cells
A major advantage of hydrogen fuel cells is that they generate electricity with minimal pollution. When hydrogen and oxygen combine to produce electricity, the by-product is simply water, making pure hydrogen fuel cells entirely carbon-free.
Other fuel cell systems can use hydrocarbon fuels like natural gas, biogas, or methanol. Since fuel cells operate through an electrochemical reaction rather than combustion, they are generally more efficient than traditional energy generation methods. Efficiency can be further improved with combined heat and power systems, which repurpose waste heat from the cell for heating or cooling.
